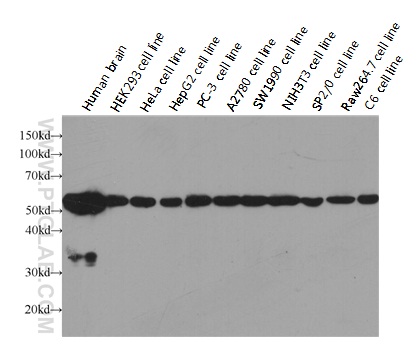
验证数据展示
经过测试的应用
| Positive WB detected in | HeLa cells, HEK-293 cells |
推荐稀释比
| 应用 | 推荐稀释比 |
|---|---|
| Western Blot (WB) | WB : 1:5000-1:50000 |
| It is recommended that this reagent should be titrated in each testing system to obtain optimal results. | |
| Sample-dependent, Check data in validation data gallery. | |
发表文章中的应用
| WB | See 172 publications below |
| CoIP | See 1 publications below |
产品信息
HRP-66031 targets Alpha Tubulin in WB, CoIP applications and shows reactivity with human, mouse, rat samples.
| 经测试应用 | WB Application Description |
| 文献引用应用 | WB, CoIP |
| 经测试反应性 | human, mouse, rat |
| 文献引用反应性 | human, mouse, rat |
| 免疫原 |
Recombinant protein 种属同源性预测 |
| 宿主/亚型 | Mouse / IgG2b |
| 抗体类别 | Monoclonal |
| 产品类型 | Antibody |
| 全称 | tubulin, alpha 1b |
| 别名 | TUBA1B, Tubulin alpha 1B chain, 1E4C11, Alpha-tubulin ubiquitous, EC:3.6.5.- |
| 计算分子量 | 50 kDa |
| 观测分子量 | 52 kDa |
| GenBank蛋白编号 | BC009314 |
| 基因名称 | Alpha Tubulin |
| Gene ID (NCBI) | 10376 |
| RRID | AB_2687491 |
| 偶联类型 | HRP |
| 形式 | Liquid |
| 纯化方式 | Antigen affinity purification |
| UNIPROT ID | P68363 |
| 储存缓冲液 | PBS with 50% glycerol, 0.05% Proclin300, 0.5% BSA, pH 7.3. |
| 储存条件 | Store at -20°C. Avoid exposure to light. Stable for one year after shipment. Aliquoting is unnecessary for -20oC storage. |
背景介绍
What is the function of alpha-tubulin?
Alpha-tubulin belongs to a large superfamily of tubulin proteins. There are a number of different subtypes that have a molecular weight of ~50kDa and are able to bind to beta-tubulin, forming a heterodimer that polymerizes to microtubules as part of the cytoskeleton. These maintain cell structure, provide platforms for intracellular transport, and are also involved in cell division.
Where is alpha-tubulin expressed?
Alpha-tubulin is highly conserved and is present in nearly all eukaryotic cells as one of the building blocks of microtubules. The ubiquitous nature of this protein has led to its common use as a control protein for many tissue types as well as highlighting the structure of the cytoskeleton.
What are the post-translational modifications of alpha-tubulin?
The function and properties of microtubules are drastically affected by the post-translational modifications undergone by tubulin, which may occur to the tubulin dimer directly or to the polymerized microtubule. For example, the first modification to be identified was detyrosination,1 as most alpha-tubulins have a tyrosine at their terminus. This process affects microtubules more than dimers and leads to patches of detyrosination along the structure, regulating protein interactions and allowing subcellular compartments to be defined.2,3 Polyglutamylation also occurs at several sites within the carboxy-terminal tails. However, to date, the most-studied alpha tubulin modification is related to acetylation of lysine 40 (K40).
Gundersen, G. G., Khawaja, S. & Bulinski, J. C. Postpolymerization detyrosination of alpha-tubulin: a mechanism for subcellular differentiation of microtubules. J. Cell Biol. 105, 251-64 (1987).
Galjart, N. Plus-End-Tracking Proteins and Their Interactions at Microtubule Ends. Curr. Biol. 20, R528-R537 (2010).
Jiang, K. & Akhmanova, A. Microtubule tip-interacting proteins: a view from both ends. Curr. Opin. Cell Biol. 23, 94-101 (2011).
实验方案
| Product Specific Protocols | |
|---|---|
| WB protocol for HRP Alpha Tubulin antibody HRP-66031 | Download protocol |
| Standard Protocols | |
|---|---|
| Click here to view our Standard Protocols |
发表文章
| Species | Application | Title |
|---|---|---|
Cell Human T-bet Governs Innate and Innate-like Adaptive IFN-γ Immunity against Mycobacteria. | ||
Mol Cancer Cell surface CD55 traffics to the nucleus leading to cisplatin resistance and stemness by inducing PRC2 and H3K27 trimethylation on chromatin in ovarian cancer | ||
Cell Immune Checkpoint Inhibition Overcomes ADCP-Induced Immunosuppression by Macrophages. | ||
Cell Stem Cell A small-molecule cocktail promotes mammalian cardiomyocyte proliferation and heart regeneration. | ||
Immunity Very-low-density lipoprotein receptor-enhanced lipid metabolism in pancreatic stellate cells promotes pancreatic fibrosis. |