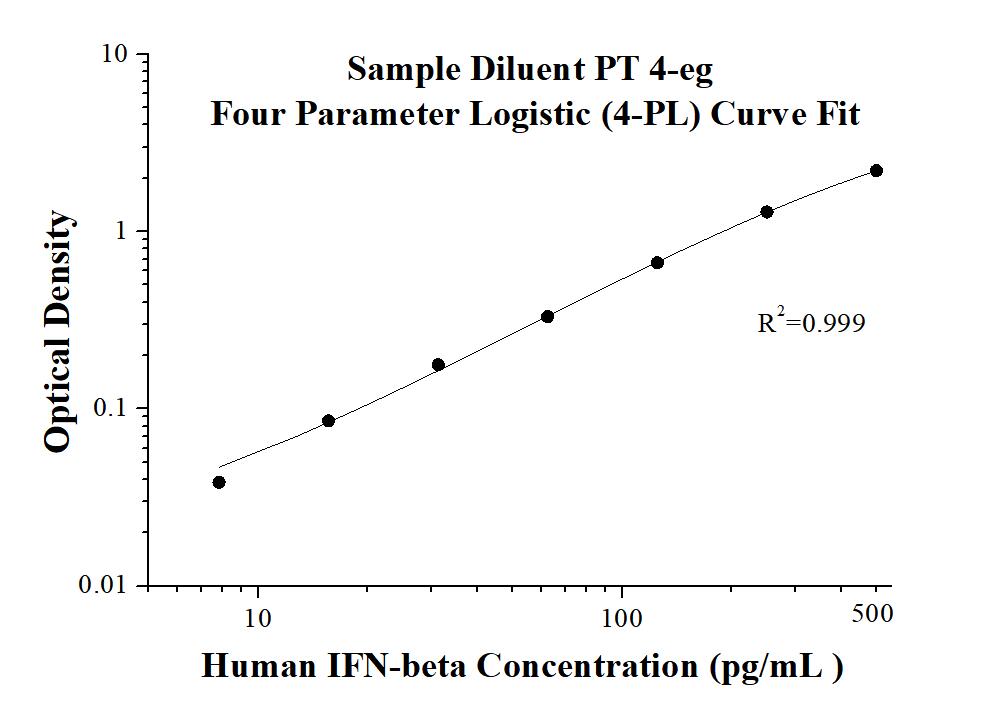
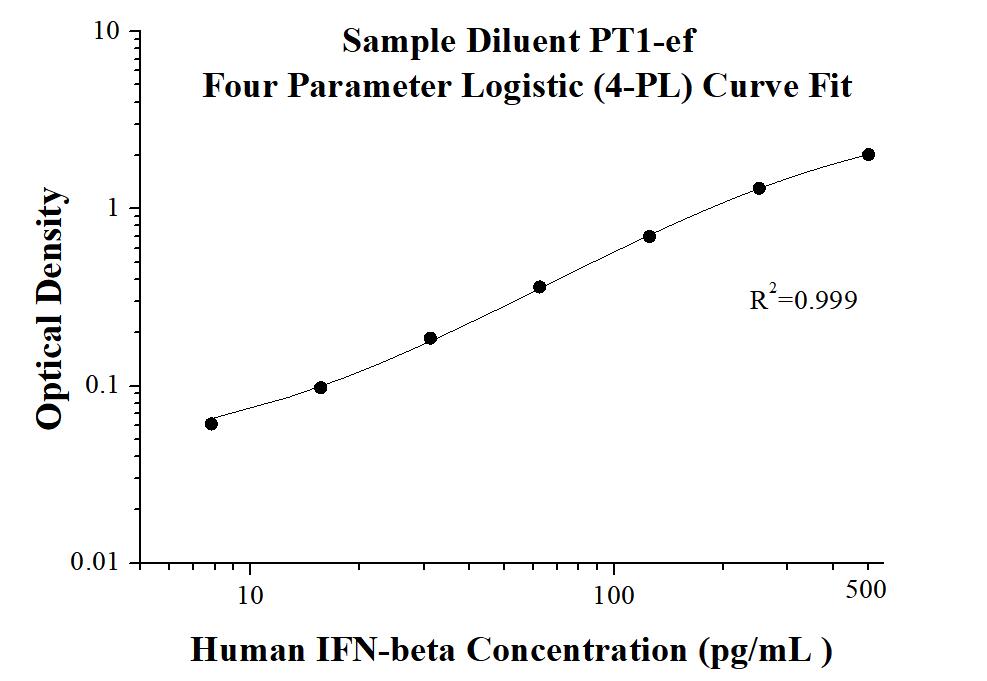
AuthentiKine™ Human IFN-beta ELISA Kit
Cat no : KE00195
Synonyms
Fibroblast interferon, IFB, IFF, IFN beta, IFNB, IFNB1, IFN-beta, IFNβ, IFN-β, Interferon beta, interferon, beta 1, fibroblast
Validation Data Gallery
Product Information
KE00195 is a solid phase sandwich Enzyme Linked-Immuno-Sorbent Assay (Sandwich ELISA). The IFN-beta ELISA kit is to be used to detect and quantify protein levels of endogenous IFN-beta. The assay recognizes human IFN-beta. An antibody specific for IFN-beta has been pre-coated onto the microwells. The IFN-beta protein in samples is captured by the coated antibody after incubation. Following extensive washing, another antibody specific for IFN-beta is added to detect the captured IFN-beta protein. For signal development, horseradish peroxidase (HRP)-conjugated antibody is added, followed by Tetramethyl-benzidine (TMB) reagent. Solution containing sulfuric acid is used to stop color development and the color intensity which is proportional to the quantity of bound protein is measurable at 450 nm with the correction wavelength set at 630 nm.
| Product name | AuthentiKine™ Human IFN-beta ELISA Kit |
| Tests | 1 X 96 well plate |
| Sample type | Serum, Plasma, Cell culture supernatants |
| Assay type | Sandwich |
| Sensitivity | 0.76 pg/mL |
| Range | 7.8-500 pg/mL |
| Reactivity | Human |
| Tested applications | Sandwich ELISA |
| Gene ID (NCBI) | 3456 |
Recovery
| Sample Type | Average | Range |
|---|---|---|
| Human Plasma | 89 | 72-107 |
| Cell culture supernatants | 84 | 78-96 |
IntraAssay
| Sample | n | mean ( pg/mL) | SD | CV% |
|---|---|---|---|---|
| 1 | 20 | 244.5 | 5.2 | 2.1 |
| 2 | 20 | 61.6 | 1.7 | 2.8 |
| 3 | 20 | 14.9 | 1.3 | 8.8 |
InterAssay
| Sample | n | mean ( pg/mL) | SD | CV% |
|---|---|---|---|---|
| 1 | 24 | 244.7 | 6.3 | 2.6 |
| 2 | 24 | 64.5 | 2.0 | 3.1 |
| 3 | 24 | 16.5 | 1.6 | 9.6 |
Background Information
Interferon beta (IFN-beta) is a cytokine that is naturally produced by the immune system in response to biological and chemical stimuli. It signals by binding to the heterodimeric type I IFN receptor composed of the IFNAR1 and IFNAR2 chains, and regulates the expression of a plethora of genes by means of the classical JAK/STAT and other pathways. Interferon beta (IFN-beta) has been shown in several clinical trials to have efficacy in multiple sclerosis. Interferon beta gene therapy is expected to become widely available for clinical use in cancer patients, and this new strategy might be extended to molecular targeting therapy, or used in combination with cell therapy or other therapies.
Properties
| Storage Instructions | All the reagents are stored at 2-8℃ for 6 months or -20℃ for 12 months. Refer to the protocol for further storage instructions. |
| Synonyms | Fibroblast interferon, IFB, IFF, IFN beta, IFNB, IFNB1, IFN-beta, IFNβ, IFN-β, Interferon beta, interferon, beta 1, fibroblast |
Publications
| Species | Sample Type | Title |
|---|---|---|
JCI Insight The proto-oncogene SRC phosphorylates cGAS to inhibit an anti-tumor immune response | ||
Virulence Cyclin-dependent kinase 5 negatively regulates antiviral immune response by disrupting myeloid differentiation primary response protein 88 self-association | ||
Int Immunopharmacol Up-regulation of HSP90α in HDM-induced asthma causes pyroptosis of airway epithelial cells by activating the cGAS-STING-ER stress pathway | ||
Dev Cell Glutamine binds HSC70 to transduce signals inhibiting IFN-β-mediated immunogenic cell death | ||
Int J Clin Exp Pathol Activation of cGAS confers PARP inhibitor resistance in ovarian cancer via the TBK1-IRF3 axis |